The ADR full form in pharmacology stands for Adverse Drug Reaction, which describes any harmful or unexpected response to a medicine taken at normal doses for prevention, diagnosis, or treatment. These reactions range from mild rashes to severe organ damage, affecting millions yearly and making safety monitoring essential in pharmacies and hospitals.
Learning about ADR full form in pharmacology helps spot issues early, preventing harm through better prescribing and patient advice. Have you seen an ADR full form in pharmacology case in practice?
Adverse Drug Reaction Definition
Adverse drug reaction refers to a sudden and unintended response to a drug at standard doses. This leads to either stopping the drug use or making variations in the doses to avoid future harm.
Unlike expected side effects, this adverse drug reaction definition stresses unpredictability and potential danger, even from approved drugs used correctly. Global bodies like WHO use this adverse drug reaction definition to track safety signals worldwide, guiding label updates or withdrawals.
Types of Adverse Drug Reactions: Main Categories
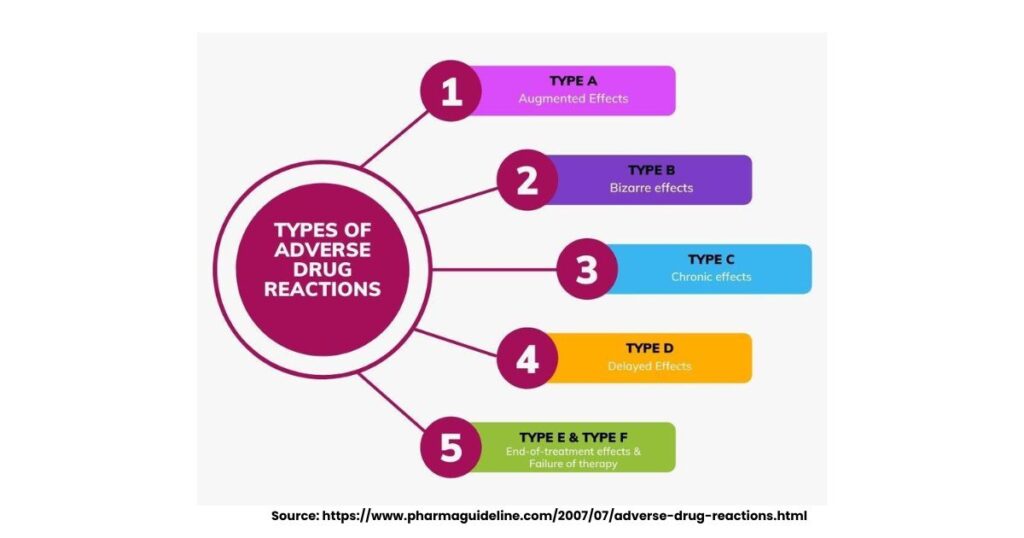
There are different types of adverse drug reactions. They can be classified by cause and predictability, helping clinicians respond fast during treatment. Type A reactions, dose-related and common (80-90%), stem from the drug’s known effects exaggerated by factors like age or kidney issues. Type B, bizarre and rare, arise unpredictably from allergies or genetics, often severe despite low doses.
Other types of adverse drug reactions include:
- Type C (chronic): Build over long use, like bone thinning from steroids
- Type D (delayed): Appear later, e.g., cancer risks or birth defects
- Type E (end): From withdrawal, like rebound hypertension off beta-blockers
- Type F (failure): Therapy fails unexpectedly, worsening condition
These types of adverse drug reactions guide monitoring in trials and practice.
Adverse Drug Reaction Examples: Common Cases
Adverse drug reaction examples bring concepts to life, showing everyday risks from familiar medicines.
For Type A, insulin overdose causes low blood sugar (hypoglycaemia) with sweating and confusion; NSAIDs like ibuprofen trigger stomach bleeding in long-term users.
Serious adverse drug reaction examples: Penicillin allergy leads to rash or anaphylaxis (Type B); chemotherapy drugs like doxorubicin cause neutropenia, raising infection odds.
Here are the primary adverse drug reaction examples by type:
- Type A: Warfarin bleeding from dose error, opioid breathing slowdown
- Type B: Amoxicillin rash or bronchospasm in allergic patients
- Type C: Corticosteroids causing cataracts over years
- Type D: Thalidomide birth defects (historical)
Who Can Report Adverse Drug Reactions?
People who can report adverse drug reactions includes doctors, nurses, pharmacists, dentists, patients, and even family. This means that anyone spotting harm from medicines plays a vital role in ensuring the patient safety. Reports go to national centres like PvPI in India or WHO global database, using forms, apps, or calls for quick entry.
Healthcare pros must report serious cases; patients encouraged for all suspicions to catch rare signals.
Who can report adverse drug reactions:
- Doctors/nurses: Serious hospital events
- Pharmacists: Community side effects
- Patients: Personal experiences
- Companies: Post-marketing data
Difference Between Adverse Drug Reaction and Adverse Drug Event
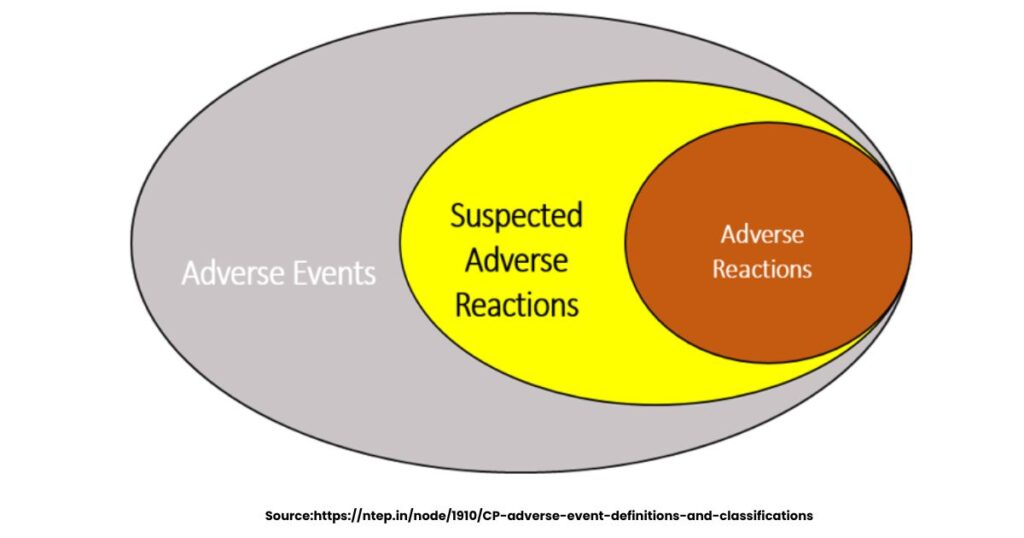
The difference between adverse drug reaction and adverse drug event centres on cause. When a person reacts to a drug at normal doses, it comes under ADR whereas when there is any harm from overdose, errors, or misuse of the drug, then this lies under ADE. ADR is a subset of ADE; ADE broader, including non-drug faults like wrong dose.
To learn more about ADE in detail, refer to this blog.
| Aspect | Adverse Drug Reaction (ADR) | Adverse Drug Event (ADE) |
| Cause | Drug itself at normal dose | Any drug-related harm (error/overdose too) |
| Predictability | Often type A/B related | Includes accidents |
| Example | Antibiotic rash | Insulin overdose hypoglycemia |
Managing and Preventing Adverse Drug Reactions
If you want to manage and prevent adverse drug reactions, you have to start with following up on the practical steps that you can complete as a part of your daily routines. Medical professionals should start with an overview of patient’s medical history to identify known allergies, previous reactions to similar medicines, or underlying conditions like kidney impairment that could amplify risks when introducing new treatments.
Here are strategies for managing and preventing adverse drug reactions effectively in everyday practice:
- Review patient history thoroughly: Check allergies, past ADRs, current drugs, and conditions like liver disease at every prescription to avoid predictable repeats.
- Begin with low doses and titrate up: Especially for high-risk groups like the elderly or children, monitor weekly in the first month for side effects like dizziness or stomach upset.
- Schedule regular monitoring: Use blood tests for drugs affecting liver or kidneys, vital signs for heart meds, and patient feedback forms for ongoing symptoms.
- Educate on warning signs: Teach recognition of common issues such as jaundice from paracetamol overuse or tremors from certain blood pressure tablets, with clear “when to call” instructions.
- Leverage interaction tools: Run checks via apps or software before adding medicines, catching overlaps like statins with grapefruit juice that spike side effects.
- Promote prompt reporting: Encourage patients, families, and staff to log even mild reactions online or via hotlines, contributing to broader safety improvements.
These steps not only reduce harm but foster stronger patient trust.
On A Final Note…
ADR full form in pharmacology alerts us to medicine risks, from adverse drug reaction definition to types of adverse drug reactions and examples. Knowing who can report adverse drug reactions and difference between adverse drug reaction and adverse drug event strengthens safety. Stay vigilant for better outcomes.
What is ADR full form in pharmacology?
Adverse Drug Reaction – harmful effect from normal drug doses.
What is the definition of adverse drug reaction?
Unintended noxious response at therapeutic doses, needing action like stop or treat.
What are the types of adverse drug reactions?
Type A (dose-related, common), Type B (idiosyncratic), C (chronic), D (delayed), E (end).
Who can report adverse drug reactions?
Doctors, pharmacists, nurses, patients – via PvPI AMCs, app, or helpline.

